Constructor Theory, Module 3: Four Laws of Thermodynamics
Constructor Theory, Module 3: Four Laws of Thermodynamics, by Conjecture Institute President Logan Chipkin.
The following is the written version of Constructor Theory, Module 3: Four Laws of Thermodynamics, by Conjecture Institute President Logan Chipkin (AI audio version on our site and embedded below). Head to Conjecture Institute’s Courses page for more. We will be adding modules across a number of courses throughout the year.
Four Laws of Thermodynamics
In Module 2, we introduced the axiomatic version of thermodynamics in the context of explaining the conflict between apparent irreversible processes and reversible dynamical laws of motion. But the laws of thermodynamics are about much more than irreversibility. In this module, we will explore the content and shortcomings of four laws of thermodynamics.
Minus-First Law: Equilibrium
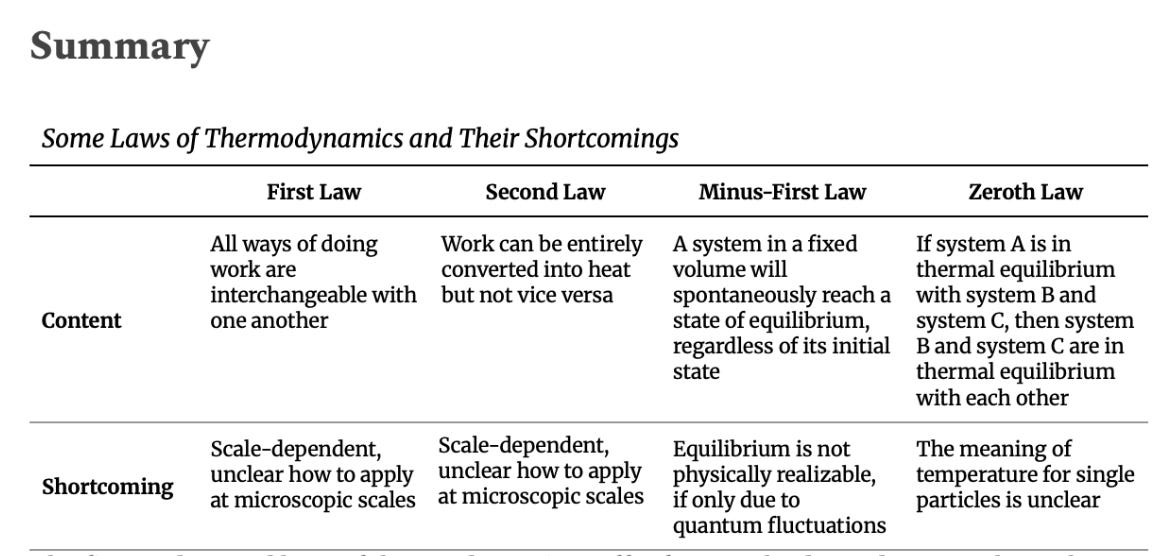
The minus-first law of thermodynamics says that a system in a fixed volume will spontaneously reach a unique equilibrium eventually, no matter its initial state. This sounds intuitive enough: a glass of initially mixed oil and water will eventually equilibrate into two distinct layers of fluid. Fumes from the kitchen thicken the air for a few minutes before spreading evenly throughout the house, merging with the much larger pool of air molecules.
What is an equilibrium state, exactly? Intuitively, it is simply a state from which a system does not deviate unless some external impetus acts on it. So we can reasonably say that a glass of separated oil and water is in equilibrium until something disturbs it—say, a cat knocking it onto a hot stove, shattering the glass and turning the oil to flame and the water to steam. A single atom can be considered in equilibrium, since its overall state can be described as orderly and globally unchanging, even though its electrons move at blazing speeds—roughly one percent the speed of light. The same goes for stars at their various stages of development—though they are certainly not in equilibrium when they are transitioning from one form to another. But, to a good approximation, our own Sun, a cooling white dwarf, and a rotating black hole are all in equilibrium—systems that are merely humming along at steady pace, unchanging at a high enough resolution scale. The minus-first law tells us that all systems, no matter how chaotic they may seem in the moment, are destined to evolve towards some particular steady state.
But if equilibrium conditions only apply at certain scales, and for arbitrary periods of time, then the minus-first law is hardly a fundamental law of physics. A truly fundamental law should hold universally—not just for glasses of water and stars, but for quantum particles, too. Worse than that, even if we somehow found an objective standard by which to measure whether or not a system is in equilibrium, quantum fluctuations guarantee that the system will deviate arbitrarily far from its equilibrium state. For example, even the tamest, stablest hydrogen molecule in empty space (that is, far from any external influences) will break apart into individuated protons and electrons somewhere in the multiverse due to quantum fluctuations.
Zeroth Law: Transitivity
The zeroth law invokes a specific kind of equilibrium known as thermal equilibrium, which is a state of affairs in which there is no net flow of heat between two or more subsystems in contact with each other. Consider a block of ice resting on a kitchen surface at room temperature. Heat will transfer from the surrounding air and kitchen surface to the ice, melting it, until the water, air, and surface are all the same temperature and heat no longer flows between them.
The zeroth law, then, states that if system A is in thermodynamic equilibrium with both system B and system C, then system B and system C are necessarily in thermodynamic equilibrium with each other. In our example, if we only knew that the water (A) was in thermodynamic equilibrium with the surrounding air (B) and with the kitchen surface (C), then the zeroth law immediately tells us that the air and the kitchen surface are also in thermodynamic equilibrium with each other.
This law is the reason why we can rely on thermometers and gives us a physical basis for the meaning of temperature—construct a partially filled vial of fluid such that the height of the fluid can be read out by an observer. Because the fluid’s height increases as it absorbs heat, we can use its height as a proxy for temperature. The fluid’s height rises when heat is transferred to it and falls when it dissipates heat. Place the vial in a reference object, such as a glass of water. The vial’s fluid will either rise (if heat flows from the reference object to the vial) or fall (if heat flows from the vial to the reference object) until the vial and the glass of water are in thermal equilibrium. The vial fluid’s height serves as a reference point against which we can compare the temperature of other objects, earning its status as a thermometer.
To be useful, a thermometer needs a fixed reference point, a physical state that is easily reproducible. The freezing point of water serves this role: place a thermometer in ice that is just barely about to melt, and label the fluid’s height as 0 degrees Celsius. We can then place the thermometer against any other object to measure its temperature relative to this standard. If the fluid in the thermometer rises above the 0 mark, the object is warmer than the freezing point of water; if it falls below, the object is colder. Thanks to the zeroth law, we know that the thermometer would read out the same height of its internal fluid for any block of ice at its freezing point—and for any other object at that same temperature. That is what makes the thermometer trustworthy: the transitivity of thermal equilibrium guarantees that equal readings mean equal temperatures.
One reason the zeroth law is problematic is that thermal equilibrium suffers from the same issues as the more general notion of equilibrium as invoked by the minus-first law. Another problem is that temperature is itself a macroscopic concept. We all understand what it means for an object such as a glass of water or heap of sand, but what is the temperature of a single particle? What does thermal equilibrium between two microscopic objects mean, if anything at all? Can we create the microscopic equivalent of a thermometer and establish a reference point in gauging microscopic temperatures as we did with macroscopic systems?
First Law: Replaceability
Informally, the first law of thermodynamics says that all ways of doing work on an adiabatically closed system (see Module 2) are equivalent. For example, whether you shake a container of water or stir it with a rod, the same mechanical work—regardless of form—will produce the same rise in temperature. Consider the thermos flask from Module 2: an insulated container of water that prevents heat from flowing to or from its surroundings. The amount of energy required to raise the water’s temperature depends on how much water the thermos contains (it takes more energy to heat two liters than one) and on how readily water absorbs energy—a property determined by its chemical composition. The stirring machine could be replaced by any other mechanical device, and the amount of work required to raise the water’s temperature would remain the same.
What about the energy source powering up the stirring machine? That, too, is infinitely replaceable. The machine might be driven by a person turning a crank, connected to the flask through a system of pulleys. Or the stirring machine could be powered by an electrical outlet, or even a child on a bike who pedals in a circle around the thermos such that each revolution causes one cycle of the stirring machine. No matter how long or complex the chain of energy sources, each can be replaced by another, and each can transfer energy to the next.
The first law tells us that the thermos of water does not ‘care’ about the path that the energy took to reach it—not the number of steps nor the form that the energy takes. The water only ‘cares’ about the total mechanical energy that is ultimately transferred to it, and that energy then raises the water’s temperature by a deterministic amount.
While energy can be transformed from one form to another, the total amount of energy is the same from beginning to end. For any complex process by which we might heat up the water, the total mechanical energy that reaches the water cannot exceed the energy put in at the start. For example, when a child gets on her bike and pushes down on the pedal to get the wheel turning—say, with ten units of energy—that is the absolute maximum amount of energy that can be transferred to the water, no matter how many intermediary steps in between the pedalling and the water. In practice, energy is lost as heat at each step along the way, so the water will always receive less than ten units of energy. The thermodynamics of efficiency give us a means by which to quantify just how efficient our various machines are (and can be) at transferring useful energy.
The first law implies that mechanical work and heat are related by a so-called equivalence relationship—both can be used to transfer energy to some target system and can be converted into one another.
Recall in Module 2 that we understood ‘mechanical work’ as being the equivalent to the displacement of a weight in a gravitational field—a simple, universal reference standard, since any form of work (stirring, shaking, cranking) can in principle be expressed as lifting a weight to some height. This works fine for macroscopic systems such as a glass of water being heated by a stirrer, as the stirrer’s motion can easily be expressed in terms of motion in a gravitational field. But what does the ‘replaceability’ feature of energy sources or the work-heat equivalence relationship imply for systems for which there is no obvious equivalent to displacement of weights in a gravitational field, such as a quantum particle? We know that it is possible to, say, raise an electron from a lower-energy quantum state to a higher-energy quantum state, but this is done by, say, the absorption of photons—and photons don’t even have mass! In short, the first law of thermodynamics is currently expressed in terms whose manifestation at microscopic scales is unclear.
Second Law: Irreversibility
We reviewed the axiomatic second law of thermodynamics in Module 2, although we did not label it as such. To briefly summarize, this version of the law states that mechanical energy can be entirely converted into heat energy (without side-effects), but the inverse is not true. A stirrer can raise a thermos of water’s temperature by transferring mechanical energy into it, but the stirrer cannot lower the water’s temperature by the same means.
The second law is scale-dependent for the same reason that the first law is. For example, what are the meaningful differences between work and heat for single particles, or for computational systems?
The first and second laws of thermodynamics suffer from scale-dependence, as they rely on concepts that have no clear analogue at microscopic scales. As we discussed in Module 2 in the context of irreversibility, the solution to these laws’ scale-dependence will entail expressing them in terms of possible and impossible transformations of physical systems.
Meanwhile, the minus-first and zeroth laws rely on notions of equilibrium, which, upon closer inspection, literally do not exist in Nature. Here, too, the solution will require us to think in terms of transformations—while equilibrium does not exist, an arbitrary sequence of transformations that keep a system stable at ever-finer scales does.
Thanks to Conjecture Institute Cofounder David Kedmey, Dirk Meulenbelt, and Edwin de Wit for valuable feedback.